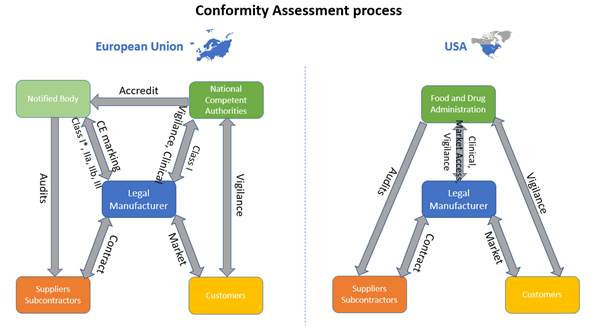
What are the principal differences between the conformity assessment process of a medical device in the USA and in the European Union? - Kvalito
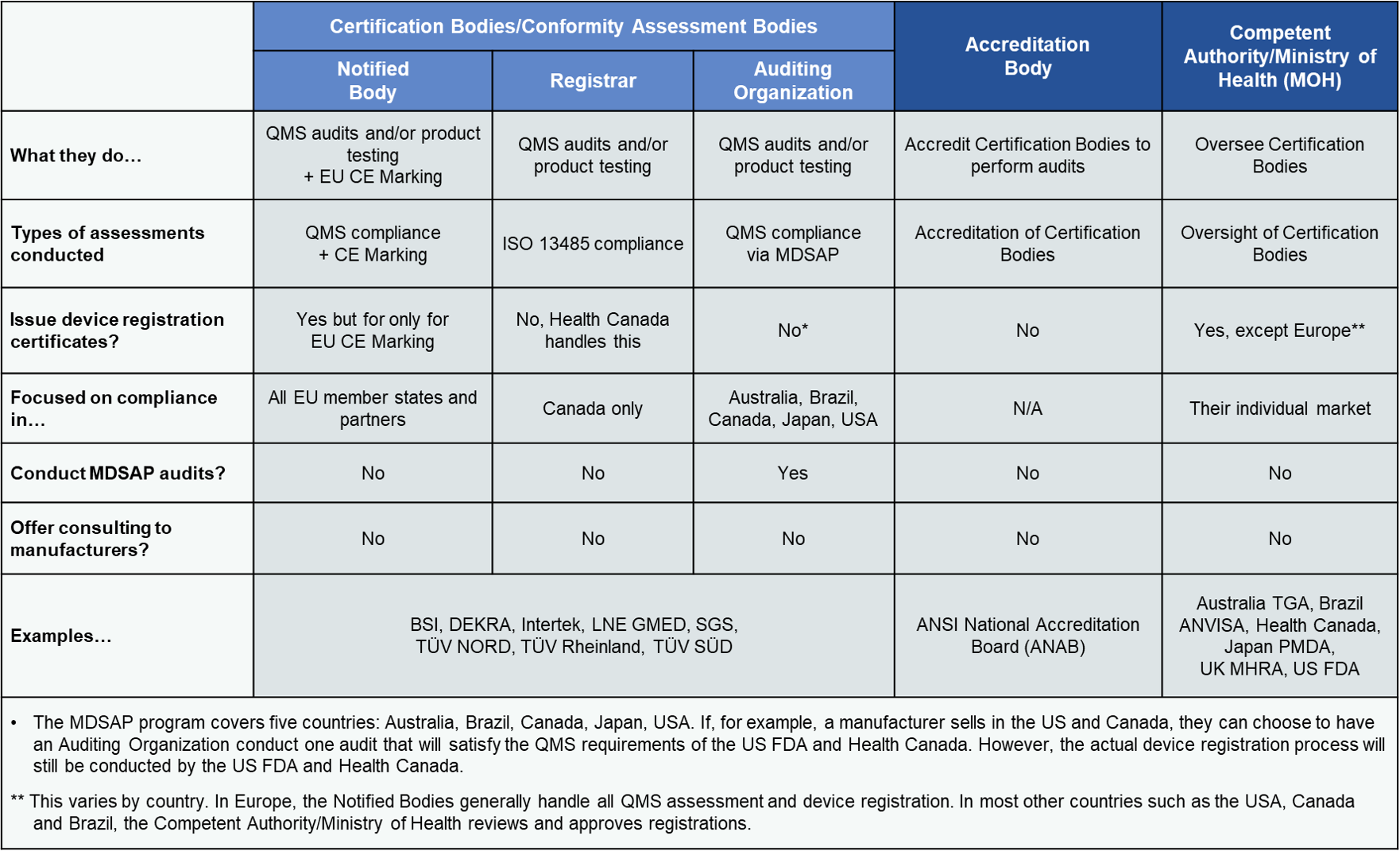
Auditing Organization (AO) versus Notified Body (NB) versus Registrar. What's the difference? – Oriel STAT A MATRIX – ELIQUENT Life Sciences Blog